A case of renal cell carcinoma diagnosed with pulmonary metastasectomy: A Case Report
Pulmonary metastasectomy
Authors
Abstract
IntroductionNearly 3% of tumors occurring in adults are renal cell carcinomas (RCCs). Approximately 20–30% of patients with renal carcinoma are diagnosed initially during the course of metastatic disease.
Case PresentationIn decreasing order of frequency, metastases are most commonly observed in the lungs, lymph nodes, liver, and bones. These patients often present with pain due to compression from the mass lesion.
ConclusionWe report a 60-year-old patient who presented to the outpatient chest clinic with pleuritic pain and was diagnosed with renal cell carcinoma following examination of a resected pleural mass.
Keywords
Introduction
Renal cell carcinoma is the most frequently seen malignant tumor of the kidney and makes up 90–95% of all kidney tumors in adults. Nearly 3% of the tumors seen in adults are renal cell carcinomas (RCCs).1 Renal cell carcinoma is the most fatal urological cancer. It has been estimated that 63,920 new cases of RCC and 13,860 deaths attributable to the disease will be reported in the United States in 2014.1
At the time of diagnosis, distant organ metastases are encountered in 30% of the patients. In patients with renal cell carcinoma, RCC metastasizes into the lungs (50–60%), bones (30–40%), liver (30–40%), and brain (5%).2 In patients with distant spread, the median survival time is nearly 10 months and the long-term survival rate is less than 5 percent.3 In this study, a case with renal cell carcinoma diagnosed when pulmonary metastasis was detected and the relevant literature were reviewed.
Case Presentation
A 60-year-old female patient consulted the outpatient clinic of chest diseases with a complaint of left flank pain. Chest radiograms of the patient, who had previously suffered from pulmonary tuberculosis, revealed diffuse nodular lesions (Figure 1a). Blood biochemistry parameters were within normal limits.
Thoracic computed tomography examination disclosed bilateral multiple hypodense soft tissue lesions with a central necrotic area in the lungs and hilar region, the largest ones measuring 5.3x3.6 cm on the right and 5.2x3 cm on the left side (Figure 1b). Because of the previous history of pulmonary tuberculosis, a lung biopsy was planned. Transthoracic fine-needle aspiration biopsy was performed, and the pathology was assessed as a result of inflammation.
Subsequently, the lesion in the right lung was excised via wedge resection performed for diagnostic and palliative purposes. The histopathological diagnosis was reported as renal cell carcinoma (Figure 2a). Abdominal tomography of the patient revealed a heterogeneous mass lesion with cystic-necrotic components originating from the right kidney and patchy areas of effacement (invasion) of fat planes between the right kidney and the liver (Figure 2b). After examinations reinforcing the diagnosis, the patient was included in a chemotherapy program in the department of medical oncology.
Discussion
Metastatic renal tumors have a poor prognosis, with a median survival time of 6–12 months and 2-year survival rates ranging between 10 and 20 percent.2 Patients generally present with complaints of abdominal pain, but they can also consult with complaints related to the organs affected by metastatic lesions. Renal cell carcinoma (RCC) is the most frequently seen urological malignancy in adults. It is most commonly observed during the 5th and 6th decades of life and is more frequently seen in men at a rate of 1.5:1. The classical triad of RCC, which consists of hematuria, flank pain, and a palpable mass, is seen in only 5–10% of patients.1 Essentially, these findings indicate diffuse disease and a poor prognosis.
Renal cell tumors can spread directly into the vena cava inferior or through venous channels into the renal vein; therefore, they easily metastasize into other organs and the lungs. In 20–30% of patients with renal tumors, the diagnosis is established when metastatic disease is detected. In 20–40% of nephrectomized patients with an indication of localized disease, metastatic disease is detected during the follow-up period.4 McLoad et al. followed 1,071 patients with extrathoracic malignancies for a period of two years.5 They found abnormalities on the chest radiograms of 163 patients, 25 of which were metastatic lesions in the hilar and mediastinal lymph nodes. The investigators reported that among extrathoracic tumors, testicular tumors (29.4%) and renal cell tumors (21.4%) were most frequently seen.5 Kutty and Varkey evaluated 46 cases of renal cell carcinoma and compared their outcomes with clinical and histopathological findings.6 During the diagnostic process, intrathoracic metastases were detected in 54% of these patients.
No renal symptoms were found in our patient. The patient had previously suffered from pulmonary tuberculosis and presented with pleuritic chest pain, which led us to search for pulmonary diseases. Though our approach to the patient was made for palliative and diagnostic purposes, interestingly, the diagnosis was established based on the examination of the metastatic lesion. Without the need for more invasive renal procedures, the quality of life of the patient improved and her survival time was prolonged. The mean survival time for these patients is 10–12 months, while the 2-year survival rate is 18–20 percent.7 Our patient was pre-emptively included in cytokine immunotherapy by the department of medical oncology. The patient is leading her routine daily life at the 18th month of her treatment without the development of further symptoms.
The applicability of metastasectomy depends on many factors, including the location, number, and excisability of the tumor, the experience of the surgeon, patient compliance, and general health state.6,7 In our patient, even though the metastasectomy was performed for diagnosis rather than with the intention to treat, establishing the diagnosis using a less invasive procedure benefited the patient despite diffuse metastases. Before systemic treatment, and despite important data suggesting a survival advantage provided by nephrectomy, a consensus has not been reached regarding the effectiveness, indications, and timing of surgical treatment in metastatic renal tumors. In a literature review of 474 patients, the rate of spontaneous regression in metastatic disease following nephrectomy alone was reported as 0.8 percent.8 In other words, nephrectomy alone is not curative in metastatic disease.
In conclusion, cases of renal cell carcinoma can present with very different clinical manifestations and demonstrate diverse clinical courses. Even in the presence of metastases, early diagnosis and treatment can increase both the survival time and the quality of life for the patient.
Declarations
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Conflict of Interest
The authors declare no conflicts of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the scientific content of the article, including the study design, data collection, analysis and interpretation, manuscript preparation, and approval of the final version of the manuscript.
References
-
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9-29. doi:10.3322/caac.21208
-
Ritchie AW, Chisholm GD. The natural history of renal carcinoma. Semin Oncol. 1983;10(4):390-400.
-
Schrader AJ, Varga Z, Hegele A, Pfoertner S, Olbert P, Hofmann R. Second-line strategies for metastatic renal cell carcinoma: classics and novel approaches. J Cancer Res Clin Oncol. 2006;132(3):137-149. doi:10.1007/s00432-005-0058-4
-
Üstün G, Turna B, Akbay K, et al. A metastatic renal cell carcinoma case diagnosed with bone metastasis. Ege Tıp Derg. 2006;45(2):145-148.
-
McLoud TC, Kalisher L, Stark P, Greene R. Intrathoracic lymph node metastases from extrathoracic neoplasms. AJR Am J Roentgenol. 1978;131(3):403-407. doi:10.2214/ajr.131.3.403
-
Kutty K, Varkey B. Incidence and distribution of intrathoracic metastases from renal cell carcinoma. Arch Intern Med. 1984;144(2):273-276. doi:10.1001/archinte.1984.00350140073011
-
Mickisch GH. Urologic approaches to metastatic renal cell carcinoma. Onkologie. 2001;24(2):122-126. doi:10.1159/000050298
-
Montie JE, Stewart BH, Straffon RA, et al. The role of adjunctive nephrectomy in patients with metastatic renal cell carcinoma. J Urol. 1977;117:272-275. doi:10.1016/s0022-5347(17)58429-3
Figures
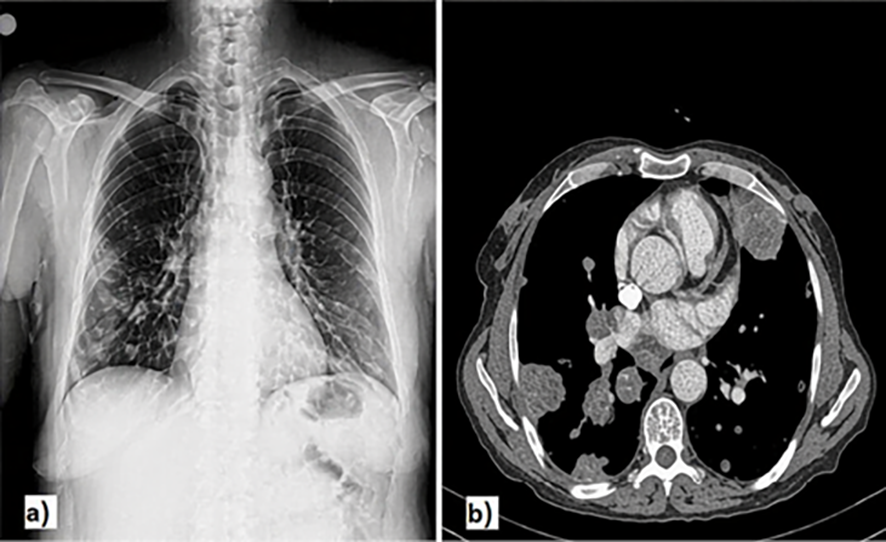
Figure 1. Diffuse nodular lesions on chest radiograms (a), On thoracic computed tomograms bilateral and multiple hypodense soft tissue lesions with central necrosis were seen on lungs and hilar regions the largest ones measuring 5.3x3.6cm on the right and 5.2x3cm on the left side (b).
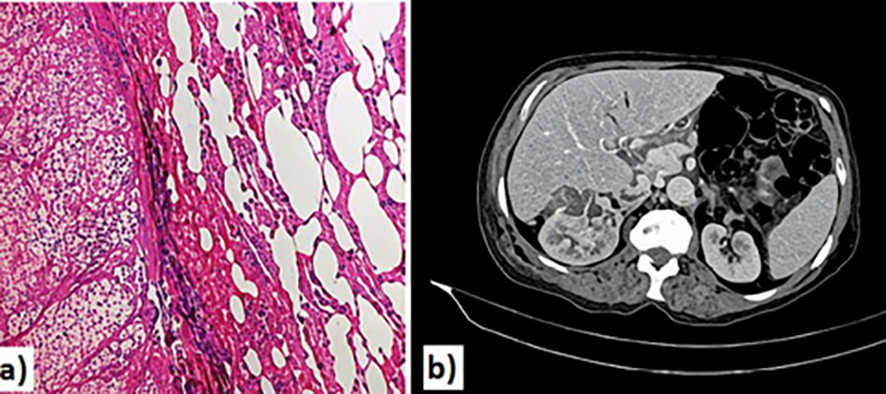
Figure 2. Metastatic lesions of clear-cell renal carcinoma in the lungs: on the left side nodular structures formed by large cells with clear cytoplasm are seen(a), A heterogenous mass with cystic-necrotic components originating from the right kidney(b).
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Selvi Asker, Ufuk Cobanoglu, Irfan Bayram, Alparslan Yavuz, Zehra Akman Ilik. A case of renal cell carcinoma diagnosed with pulmonary metastasectomy: A Case Report. Eu Clin Anal Med 2015;3(3):30-32. doi:10.4328/ECAM.59
- Received:
- October 9, 2015
- Accepted:
- October 19, 2015
- Published Online:
- October 19, 2015
- Printed:
- October 19, 2015
