Management of carotid body tumors: a report of first 8 cases
Management of carotid body tumors
Authors
Abstract
AimCarotid body tumors (CBT) are rare vascular neoplasms arising from the paraganglia. CBT resection is still a surgical challenge and is associated with neurologic and vascular omplications. In our study, we would like to report our first experiences in the surgical management of CBTs and compare our results with the literature.
MethodsSeven patients were identified with a total of 8 CBTs between June 2012 and June 2014. 5 (71.4%) of the patients were male and 2 (28.9%) of the patients were female with a mean age of 38-years (range 34-46 years). Tumors were graded according to the Shamblin classification as one of the patients was type I, five of them were type II and two patient was type III. In patients with Shamblin type I and II, careful dissection of the tumor was conducted by means of thermal cautery in the subadventitial plane. The patient with Shamblin type III, after inserting an intraluminal shunt to maintain cerebral perfusion, 2 cm distal portion of the Common Carotid Artery (CCA) and 3.5 cm proximal portion of the Internal Carotid Artery (ICA) were excised together with the mass leaving 0.5 cm of healthy tissue as a border. External Carotid Artery (ECA) was ligated and a saphenous graft was interpositioned between the CCA and ICA.
ResultsPatient who underwent surgical management for staged bilateral CBT (right side was Shamblin type II, left side was Shamblin type I), developed fascial paralysis, difficulty in swallowing, hoarseness of voice and malignant hypertension after the first operation which was treated medically. Following the second operation, malignant hypertension, fascial paralysis and difficulty in swallowing developed again. The same therapeutic strategy was followed and the patient’s complaints were recovered after 3 months.
In one patient with right side Shamblin type II CBT, localized haematoma was developed which did not require a surgical approach.
ConclusionSurgical and diagnostic management of CBTs is challenging. We reported the first 8 cases of CBTs operated in our clinic. Our first experience were found consistent results when compared to other modern case series
Keywords
Introduction
Carotid body tumors (CBT), which are known as paragangliomas, are rare vascular neoplasms arising from the paraganglia. In the head and neck, paraganglia are located in close association with the paranervous system along the arterial vasculature and cranial nerveous system. The carotid body originates from the third branchial arch mesoderm and from ectodermal derived neural crest lineage.1,2 It is located posteriorly within the adventitia at the bifurcation of the common carotid artery. It is innervated by the glossopharyngeal nevre1 and has a very rich blood supply deriving from vasa vasorum, vertebral artery and the branches of external carotid artery.3 Its function, embryology and histology is similiar with other glands of neural crest and plays an important role in cardiorespiratory performance. It responds to hypoxia and sends signals to respiratory control center to augment ventilation, heart rate and blood pressure.
They are generally benign, slow-growing and asymptomatic tumors and the incidence is between 0.06-3.3/100.000 in literature.4,5 Following the diagnosis, early surgical management is the only curative treatment to prevent the invasion and/or local mass affect on the adjacent vascular and neural tissues. However, CBT resection is still a surgical challenge and is associated with neurologic and vascular complications. In our study, we would like to report our first experiences in the surgical management of CBTs and compare our results with the literature.
Materials and Methods
Seven patients were identified with a total of 8 CBTs between January 2013 and March 2014. 5 (71.4%) of the patients were male and 2 (28.9%) of the patients were female with a mean age of 38-years (range 34-46 years). Five tumors were located on the right side and three tumors were located on the left side. One patient (14.2%) had bilateral CBT. Tumors were graded according to the Shamblin classification as one of the patients was type I, five of them were type II an two patient was type III. The most common symptom was a slowly growing lateral neck mass. Other symptoms included pain at pressure on the mass and thrill or pulse-like sensation. None of the patients had neurologic complaints. Physical examination revealed a pulsatile lateral neck mass loctaed below the mandibular angle exhibiting medial and lateral mobility but immobile on the cephalocaudal plane.
Dupplex ultrasound scanning, and computed tomography (CT) were performed in all patients. After confirming the diagnosis, magnetic resonance arteriography (MRA) of the head and neck was arranged. A low T1 signal intensity with a high T2 intensity signal was noted in all cases. Figure 1 shows the Magnetic resonance imaging (MRI) and Figure 2 shows the MRA images of the patient with bilateral CBTs.
Green Arrows Show the Carotid Bbody TumorsDigital subtraction angiography (DSA) was performed in all patients. Figure 3 shows the DSA images of the same patient. Urine catecholamine levels were measured in all cases. No patients underwent preoperative embolization. All surgeries were conducted under general anesthesia via cervical approach on the anterior border of the sternocleidomastoid muscle. Careful dissection was deeply extended to expose the carotid vessels characteristically splayed by CBT. Figure 4 shows the intraoperative photograph of an exposed CBT in the bifurcation of the carotid artery prior to dissection of the tumor.
Vascular loops were applied on ECA, ICA and CCA along with any major branches to control eventual vascular complications. The hypoglossal nevre was dissected and seperated from the surgical plane. The vagus, glossopharyngeal, ansa cervicalis, recurrent laryngeal nerves were also protected. In shamblin I and II tumors, a periadventitional caudal-cranial dissection was performed along the relatively avascular white-line plane using bipolar coagulation. The ICA is carefully dissected from the superior egde of the tumor. First the lateral face of the ICA is liberated followed by the medial face, and the vascular dissection progressed below the bifurcation in the subadventitial plane. Figure 5 shows an intraoperative photograph of the same patient after the resection of the CBT.
The ECA was preserved where possible but in three patients, ECA was ligated. Blood flow was not ceased in these patients during the operation. In one patient with Shamblin type III CBT, tumor could not be enucleated alone. After inserting an intraluminal shunt to maintain cerebral perfusion, 2 cm distal portion of the CCA and 3.5 cm proximal portion of the ICA were excised together with the mass leaving 0.5 cm of healthy tissue as a border. ECA was ligated and a saphenous graft was interpositioned between the CCA and ICA. All masses were confirmed pathologically as CBTs
Results
The female patient who underwent surgical management for staged bilateral CBT (right side was Shamblin type II, left side was Shamblin type I) developed facial paralysis, difficulty in swallowing, hoarseness of voice, and malignant hypertension after the first operation. Hypertension was controlled by administration of intravenous nitroglycerin and diltiazem, followed by oral diltiazem (3x60 mg). At the postoperative 14th day, the anti-hypertensive therapy was stopped.
In the immediate postoperative course, 1 mg/kg prednisolone and mannitol treatment was initiated for neurological morbidities and continued for 3 days. At the 2nd-month follow-up, the patient had no neurologic complaints. After evaluating the vocal and swallowing functions, the second intervention was arranged for the left side. Following the second operation, malignant hypertension, facial paralysis, and difficulty in swallowing developed again. The same therapeutic strategy was followed, and the patient’s complaints recovered after 3 months.
In one patient with right-side Shamblin type II CBT, a localized haematoma developed which did not require a surgical approach.
No other local (wound infection, cervical nerve injury, vein graft occlusion) or systemic (stroke, Transient Ischemic Attack, Horner’s syndrome) complications developed. No evidence of local or distant metastasis was observed during the follow-up of the patients.
Discussion
CBTs are rare and invaribly benign but malignant forms can be seen occasionally. CBT is usually a non-funtioning tumor but a number of neurochemicals such as tyrosine hdroxylase, nitric oxide synthase, insulin-like growth factor, norepinephrine, epinephrine, dopamine, acethylcholine, endothelin, vasoactive intestinal polypeptide or substance P have been isolated from normal or abnormal carotid bodies.6 Carotid body is one of the first responders to low PaO2 and /or reduced O2 content of inspired air. the gland responds the alterations in PaCO2 and pH as well.7 As a result, chronic hypoxemia, that is typified by diseases accompanied by very low PaO2 like sleep apnea or by dwelling at high altitudes (above 1500 m), is a stimulus for hypertrophy and hyperplasia.8 Genetic factors are also thought to be involved in the pathogenesis of this disease. Fisch et al., reported a linkage to chromosomes 11q23, 11q13 and 1q21, with a mutation in the succinate dehydrogenase subunit (SDH) B, C or D.9 The proportion of heritable cases range from 7% to 35% in literature.6,7,10,11
CBT has a higher occurance rate in females with an approximate ratio of 1:1.9.4 The reason for higher female preponderance is unknown but may be related to hormonal changes during menstruation and pregnancy. In our study 71.4% of the patients were male but the number of the patients was too small to compare with other studies.
CBTs are often asymtomatic and most patients present with neck asymmetry or a neck lump. Pulse-like vibratory sense, pain at pressure in the mass site and pressure symptoms such as neck pain, stridor, hoarseness, dysphagia, odynophagia, dysphonia, jaw stiffness, sore throat or cranial nerve palsy.4,11 Symptoms mediated by endocrine changes are rare12 and did not ocur in our study. Generally the mass can be moved in the lateral plane but not the cephalocaudal plane because of its adherence to the carotid vasculature; so called the Fontaine sign.13
According to the relation to the carotid vessel wall which is determinded by imaging studies, Shamblin et al. classified CBTs into three types as Type I: CBTs that are relatively free of involvement of the vessel walls, tumor size < 5 cm., no widened carotid bifurcation and easily resectable; Type II: CBTs that are attached to the vessel wall but without encasement; TypeIII: CBTs that are intramural and do encase ICA and ECA as well as regional nerves, tumor size > 5 cm with widened carotid bifurcation.14
Color Doppler sonography is our first choice for diagnostic investigation because it is noninvasive and depicts growth at carotid bifurcation causing splaying of the ICA and ECA an their branches due to displacement by mass (Lyre sign) and helps to evaluate the hypervascularity. CT is useful in demonstrating the diffusion of the tumor to adjacent structures. MRA of the head and neck can provide all the information necessary to operate if the tumor is solitary and the family history is negative. DSA is still the gold standart for the final diagnosis.15 It provides us diagnostic information about vascular anatomy, carotid arterial neovascularization and Shamblin’s classification.
Despite its benign behaviour, surgical resection is the treatment of choice after diagnostic evaluation. We preferred careful subadventitial resection which was described by Gordon Taylor for the surgical management of Shamblin type I and II CBTs.16 One patient with Shamblin Type III CBT required the resection of the ECA and ICA with the mass followed by subsequent vascular reconstruction with a saphenous vein graft. It is not essential to ligate the ECA and careful use of bipolar cautery and ligation of all feding vessels before dissection can reduce blood loss. However, some authors advise the ligation of ECA to devascularise the feding vessels of the tumour.17,18 We did not routinely performed elective ligation of the ECA except three cases (37.5%) where it was necessary for haemostasis.
Preoperative arterial embolisation for highly vascular CBTs with polyvinyl alcohol or gelfoam is recommended by some authors.18,19 They mainly claim that the procedure reduces blood loss and surgery duration, leads potential reductions in tumour size and vascularity and facilitate surgical resection. However postponing the surgery can result in revascularization edema associated with a localised inflammatory response which can complicate the dissection.10,20 Furthermore, supraselective embolization is difficult, especially in large CBTs, and this time-consuming process is associated with a real risk of distal migration of the embolisation medium. The stroke incidence has been reported as >10% after embolisation.21,22 As a result, we did not apply preoperative embolisation in any cases due to conflicting results in literature.
Surgical management of bilateral tumors requires more attention and should not be treated in one surgical intervention because of the risk of neurologic injury. One patient presented with a bilateral CBT (14.2%). Cranial nevre injury is the most common complication (range between 32-44%) after the excision of CBTs due to the close localization to facial, vagus, hypoglossal and glossopharyngeal nerves.18 Especially vagus is more affected because of occasional nevre retraction or sacrifice to facilitate tumor excision.
Radiotherapy is another contraversial issue. It is recommended for regional lymph node metastasis of CBTs which are large in size, recurrent and malign.23 Since CBTs grow slowly, with a median doubling time of 4.2 years, the indications should be limited to only to nonoperable patients. Furthermore, radiotherapy carries a risk of subsequent radiation-induced malignant degeneration, ageusia, xerostomia and skin rash.10,24
Conclusion
Surgical and diagnostic management of CBTs is challenging. We reported the first 8 cases of CBTs operated in our clinic. Our first experience is comparable with other modern case series reports.
Declarations
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed Consent
Informed consent was obtained from all participants.
Conflict of Interest
The authors declare no conflicts of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the scientific content of the article, including the study design, data collection, analysis and interpretation, manuscript preparation, and approval of the final version of the manuscript.
References
-
Mitchell RO, Richardson JD, Lambert GE. Characteristics, surgical management, and outcome in 17 carotid body tumors. Am Surg. 1996;62(12):1034-1037.
-
Williams MD, Phillips MJ, Nelson WR, Rainer WG. Carotid body tumor. Arch Surg. 1992;127(8):963-968. doi:10.1001/archsurg.1992.01420080097016
-
Najibi S, Terramani TT, Brinkman W, Thourani VH, Smith RB, Lumsden AB. Carotid body tumors. J Am Coll Surg. 2002;194:538-539. doi:10.1016/s1072-7515(02)01131-6
-
Sajid MS, Hamilton G, Baker DM. A multicentre review of carotid body tumour management. Eur J Vasc Endovasc Surg. 2007;34(2):127-130. doi:10.1016/j.ejvs.2007.01.015
-
Plukker JT, Brongers EP, Vermey A, Krikke A, van den Dungen JJ. Outcome of surgical treatment for carotid body paraganglioma. Br J Surg. 2001;88(10):1382-1386. doi:10.1046/j.0007-1323.2001.01878.x
-
Knight TT Jr, Gonzalez JA, Rary JM, Rush DS. Current concepts for the surgical management of carotid body tumor. Am J Surg. 2006;191(1):104-110. doi:10.1016/j.amjsurg.2005.10.010
-
Şanlı A, Oz K, Ayduran E, Aydin S, Altin G, Eken M. Carotid body tumors and our surgical approaches. Indian J Otolaryngol Head Neck Surg. 2012;64(2):158-161. doi:10.1007/s12070-011-0461-x
-
Lahiri S, Di Giulio C, Roy A. Lessons from chronic intermittent and sustained hypoxia at high altitudes. Respir Physiol Neurobiol. 2002;130:223-233. doi:10.1016/s0034-5687(01)00343-7
-
Fisch JH, Klein-Weigel P, Biebl M, Janecke A, Tauscher T, Fraedrich G. Systematic screening and treatment evaluation of hereditary neck paragangliomas. Head Neck. 2007;29(9):864-873.
-
Makeieff M, Raingeard I, Alric P, Bonafe A, Guerrier B, Marty-Ane C. Surgical management of carotid body tumors. Ann Surg Oncol. 2008;15(8):2180-2186. doi:10.1245/s10434-008-9977-z
-
O’Neill S, O’Donnell M, Harkin D, Loughrey M, Lee B, Blair P. A 22-year Northern Irish experience of carotid body tumours. Ulster Med J. 2011;80(3):133-140.
-
Zhu J, Han D, Zhou J. Retrospective analysis of the diagnosis and treatment of 11 cases of carotid body tumor. Lin Chuang Er Bi Yan Hou Ke Za Zhi. 2005;19:817-819.
-
Sari M, Serin GM, Ozdemir N, Inanli S, Tutkun A. Karotid cisim tumorlerinde tedavi yaklasimlarimiz. KBB Forum. 2006;5(3):113-117.
-
Shamblin WR, ReMine WH, Sheps SG, Harrison EG. Carotid body tumor (chemodectoma). Am J Surg. 1971;122(6):732-739. doi:10.1016/0002-9610(71)90436-3
-
Ma D, Liu M, Yang H, Ma X, Zhang C. Diagnosis and surgical treatment of carotid body tumor: a report of 18 cases. J Cardiovasc Dis Res. 2010;1(3):122-124. doi:10.4103/0975-3583.70905
-
Gordon-Taylor G. On carotid body tumours. Br J Surg. 1940;28:163-172. doi:10.1002/bjs.18002811003
-
Koskas F, Vignes S, Khalil I, et al. Carotid chemodectomas: long-term results of subadventitial resection with deliberate external carotid resection. Ann Vasc Surg. 2009;23(1):67-75. doi:10.1016/j.avsg.2008.01.015
-
Persky MS, Setton A, Niimi Y, Hartman J, Frank D, Berenstein A. Combined endovascular and surgical treatment of head and neck paraganglioma—a team approach. Head Neck. 2002;24(5):423-431. doi:10.1002/hed.10068
-
Davidovic LB, Djukic VB, Vasic DM, Sindjelic RP, Duvnjak SN. Diagnosis and treatment of carotid body paraganglioma: 21 years of experience at a clinical centre of Serbia. World J Surg Oncol. 2005;3(1):10. doi:10.1186/1477-7819-3-10
-
LaMuraglia GM, Fabian RL, Brewster DC, et al. The current surgical management of carotid paragangliomas. J Vasc Surg. 1992;15(6):1038-1045.
-
Litle VR, Reilly LM, Ramos TK. Preoperative embolization of carotid body tumors: when is it appropriate? Ann Vasc Surg. 1996;10(5):464-468. doi:10.1007/bf02000594
-
Baskoyannis KC, Georgopoulos SE, Klonaris CN, et al. Surgical treatment of carotid body tumors without embolization. Int Angiol. 2006;25(1):40-45.
-
Gerosa M, Visca A, Rizzo P, Foroni R, Nicolato A, Bricolo A. Glomus jugulare tumors: the option of gamma knife radiosurgery. Neurosurgery. 2006;59(3):561-569. doi:10.1227/01.neu.0000228682.92552.ca
-
Hallett JW Jr, Nora JD, Hollier LH, Cherry KJ Jr, Pairolero PC. Trends in neurovascular complications of surgical management for carotid body and cervical paragangliomas: a 50-year experience with 153 tumors. J Vasc Surg. 1988;7(2):284-291. doi:10.1067/mva.1988.avs0070284
Figures
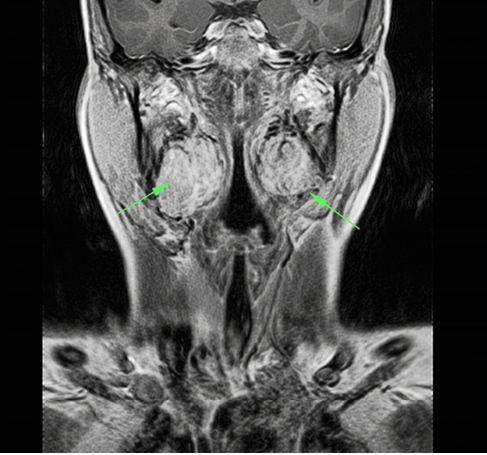
Figure 1. Magnetic resonance imaging study of the patient with bilateral carotid body tumor

Figure 2. Magnetic resonance arteriography image of the patient with bilateral carotid body tumor
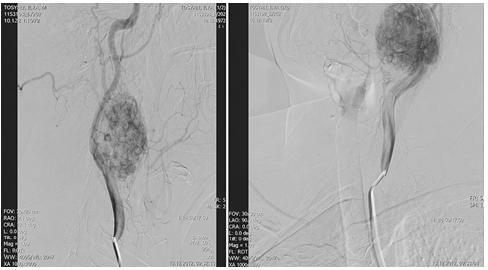
Figure 3. Digital Subtraction Angiography images of the patient with bilateral carotid body tumor
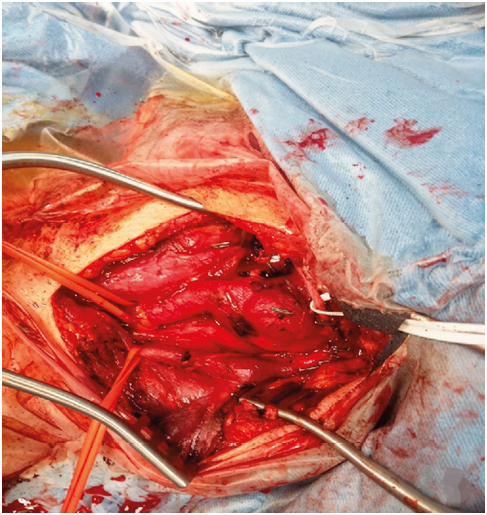
Figure 4. Intraoperative photograph of an exposed CBT in the bifurcation of the carotid artery prior to dissection of the tumor
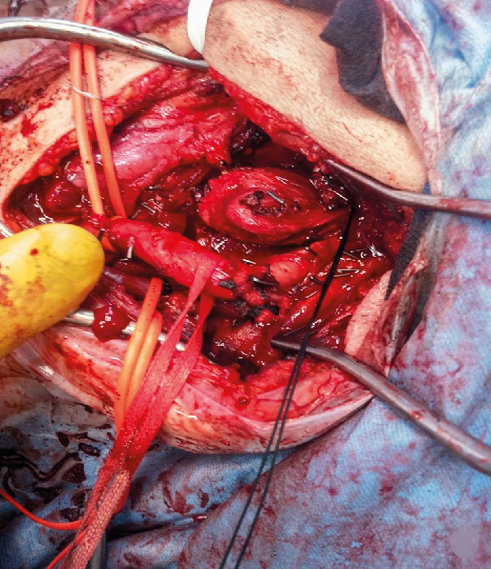
Figure 5. Intraoperative photograph of the same patient after the resection of the CBT
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Kemal Korkmaz, Hikmet Selçuk Gedik, Ali Baran Budak, Serhat Bahadır Genç, Naim Boran Tümer, Kerim Çağlı. Management of carotid body tumors: a report of first 8 cases. Eu Clin Anal Med 2014;2(3):19-22. doi:10.4328/ECAM.35
- Received:
- September 13, 2014
- Accepted:
- September 17, 2014
- Published Online:
- September 17, 2014
- Printed:
- September 17, 2014
