Serum copeptin levels and its relation with other inflammatory markers in acute exacerbation of COPD
Acute exacerbation of COPD and copeptin
Authors
Abstract
AimCopeptin has been introduced as an inflammation marker in recent years. The role of copeptin as an inflammation marker in the short-term period of Acute Exacerbation of COPD (AECOPD) has not yet been well demonstrated. We aimed to investigate the course of copeptin and its correlation with other inflammatory markers in AECOPD during hospitalization.
MethodsForty-six AECOPD patients (42 male, 4 female) were included in the study. Blood leukocyte count, C-Reactive Protein (CRP), Brain Natriuretic Peptide (BNP), and copeptin levels were measured on days 0, 3, 7, and 14, respectively.
ResultsThe mean age of the patients was 65 \pm 10 years. Copeptin median levels were 129.8, 170.8, 235.6, and 338.4 pmol/L on days 0, 3, 7, and 14, respectively. CRP median levels were 3.42, 1.65, 0.73, and 1.15 mg/L, respectively. Serum BNP median values were 97, 88, 43, and 2.5 U/L, respectively. Copeptin was not significantly associated with CRP, leukocyte count, or BNP during the study period.
ConclusionIn this study, we observed that serum copeptin levels increased, in contrast to the decrease in leukocyte, CRP, and BNP levels during AECOPD. As no correlation was observed between copeptin and other markers, we believe that copeptin may not be an early inflammation marker of AECOPD, contrary to other reports in the literature.
Keywords
Introduction
Chronic Obstructive Pulmonary Disease (COPD) is a condition associated with an increasing rate of morbidity and mortality.1 The clinical prognosis is worse in patients with COPD during the exacerbation period versus the stable period. Exacerbations are the most common indicators of treatment failure and disease progression.2
With reference to both clinical findings and laboratory results, differential diagnosis of the stable and exacerbated period at a late stage of the disease is very difficult. Lower Respiratory Tract Infection (LRTI) is the most common cause of exacerbations.2,3 However, heart failure is also common among patients with Acute Exacerbations of COPD (AECOPD) (20–40%); for this reason, it is generally problematic to differentiate pure AECOPD from heart failure. Traditional biomarkers, including fever, white blood cells count, and serum C-Reactive Protein (CRP) level, have been reported to be unreliable for assessment of the mortality rate and severity of the disease.4
B-Type natriuretic peptide (BNP) is a neurohormone elevated in states of increased ventricular wall stress. While it has been established to be a good indicator of heart failure, it can also be elevated in a wide variety of clinical settings with or without Congestive Heart Failure (CHF), and in non-cardiac diseases such as COPD and pulmonary embolism. Most dyspnea patients with heart failure have BNP values higher than 400 pg/mL, whereas left ventricular dysfunction without exacerbation, pulmonary embolism, or cor pulmonale should be considered in dyspnea patients with plasma BNP concentrations between 100 and 400 pg/mL.5
It has been shown that BNP levels in AECOPD can be directly correlated with common markers of infection, such as CRP and procalcitonin. BNP levels may, therefore, accurately reflect the presence and reveal the severity of the most prominent prognostic factors in AECOPD.6
Thus, one approach in estimating the severity and predicting the outcome of the exacerbation of COPD is the use of novel biomarkers.2 Arginine Vasopressin (AVP) is a hormone increasing in hypotensive, hypoxic, hyperosmolar or acidotic stimuli and infectious conditions. Its instability makes it difficult to routinely take reliable measurements.
Copeptin is a more stable C-terminal part of AVP and directly reflects levels of vasopressin. It can be easily measured during clinical routine and effectively mirrors vasopressin levels.7,8,9,10 It then seems reasonable to speculate that the diverse secretion sites and the smaller amount of heart-specific release mechanisms of copeptin compared to BNP may at least partly explain the different predictive potential of these biomarkers in AECOPD.6
A recent study reported copeptin to be of interest as a prognostic biomarker for short-term and long-term prognoses in patients with AECOPD requiring hospitalization.11
The aim of the current study was thus to investigate the usefulness of copeptin in assessing in-hospital prognosis of AECOPD and its correlation with serum CRP and BNP levels during the hospitalization period.
Materials and Methods
Study Design and Patient SelectionPatients (age range: 40-75) consecutively admitted to Yedikule Hospital For Chest Diseases and Thoracic Surgery Training and Research between 01.09.2008-31.12.2008 with a preliminary diagnosis of AECOPD were included into the study. The diagnosis of COPD and exacerbation was based on the definition of the American Thoracic Society.1 Inclusion criteria at entry were a change in the patient’s baseline dyspnea, cough, and/or sputum that is beyond normal day-to-day variations which is acute in onset, and a change in regular medication in a patient with underlying COPD and additionally smoking history more than 20 pack-years and no history suggestive of asthma. Patients with a history of renal failure, hepatic failure, recent Acute Myocardial Infarction (AMI) (in the past one year), active tuberculosis or cancer, immunodeficiency, suspected asthma or Cystic Fibrosis (CF), or large infiltrations in the x-ray were excluded from the study.
Clinical and Laboratory AssessmentPatients were evaluated in terms of age, sex, dyspnea, cough, sputum characteristics, the amount of sputum, cigarette smoking status (smokers, non-smokers, quitters), number of smoking pack-years, biomass exposure status and duration, number of referrals to an emergency department, length of stay in the intensive care unit, physical examination findings, axillary body temperature, and presence of Anthonisen criteria.12 Chest x-ray, Arterial Blood Gas (ABG) analysis, serum hemoglobin and hematocrit levels, platelet and eosinophil count, and biochemical parameters (urea, creatinine, ALT, AST, total protein, albumin, total bilirubine, direct and indirect bilirubine, total cholesterol, LDL-cholesterol, HDL-cholesterol, CRP, Brain Natriuretic Peptide (BNP), sedimentation rate, d-dimer), sputum gram staining and history of antibiotic use due to acute exacerbation at baseline or in the past six weeks were all noted. Blood complete blood counts, serum Copeptin, CRP, and BNP levels were evaluated on days 0, 3, 7 and 14 post-admission. Patient’s sputum was analyzed by gram staining and nonspecific culture.
Signed informed consent forms were obtained from all patients.
CopeptinCopeptin was analyzed by commercial ELISA kits (Phoenix Pharmaceuticals Inc, Belmont, CA, USA).
Preparation of SerumFor the preparation of serum, 4-mL blood samples were collected in tubes containing EDTA and shaken a few times for anticoagulation. Samples were transferred into biochemistry tubes containing Aprotinin (0.6 TIU/mL) to inhibit proteinase activity and shaken. Tubes were centrifuged at 1600 x g for 15 minutes. Plasma was stored at -20° C for two months and analyzed in the laboratory supplying the analysis kit.
ELISAThe immuno plate in this kit is pre-coated with a secondary antibody with nonspecific binding sites blocked. The secondary antibody is able to bind to the Fe fragment of the primary antibody (peptide antibody) whose Fab fragment will be competitively bound by both biotinylated peptide and peptide standard or targeted peptide in samples. The biotinylated peptide interacts with streptavidin-horseradish peroxidase (SA-HRP), catalyzing the substrate solution. The intensity of the yellow is directly proportional to the amount of biotinylated peptide SA-HRP complex, but inversely proportional to the amount of peptide in standard solutions or samples due to competitive binding of the biotinylated peptide with the standard peptide or samples to the peptide antibody (primary antibody). A standard curve of known concentration can be established accordingly, with an unknown concentration in samples determined by extrapolation to this standard curve.
CRP was analyzed using the standard turbulometric method. BNP was analyzed using standard electro-chemilluminescence immunoassay.
Radiological AnalysisDigital chest x-ray was performed for all patients.
Statistical AnalysisStatistical analyses were performed using a statistical software program (SPSS for Macintosh, Gradpack Rel. 17.0.0, SPSS Inc., Chicago, IL, USA). Qualitative measurements were defined in reel numbers and percentages. Quantitative variables were defined in mean ± standard deviation (min-max) for normal distribution, but in median values in the absence of normal distribution. The ANOVA test was performed to compare groups in normal distribution, while the Kruskal-Wallis test was used in the non-normal distribution. Paired t-tests were used to compare the dependent variables in normal distribution, whereas the Mann Whitney and Wilcoxon Rank sum tests were used in the non-normal distribution. (Chi-square test was also used to compare qualitative variables between the groups). Pearson’s correlation test was performed to analyze the correlation between the variables. p<0.05 was considered to be statistically significant.
Results
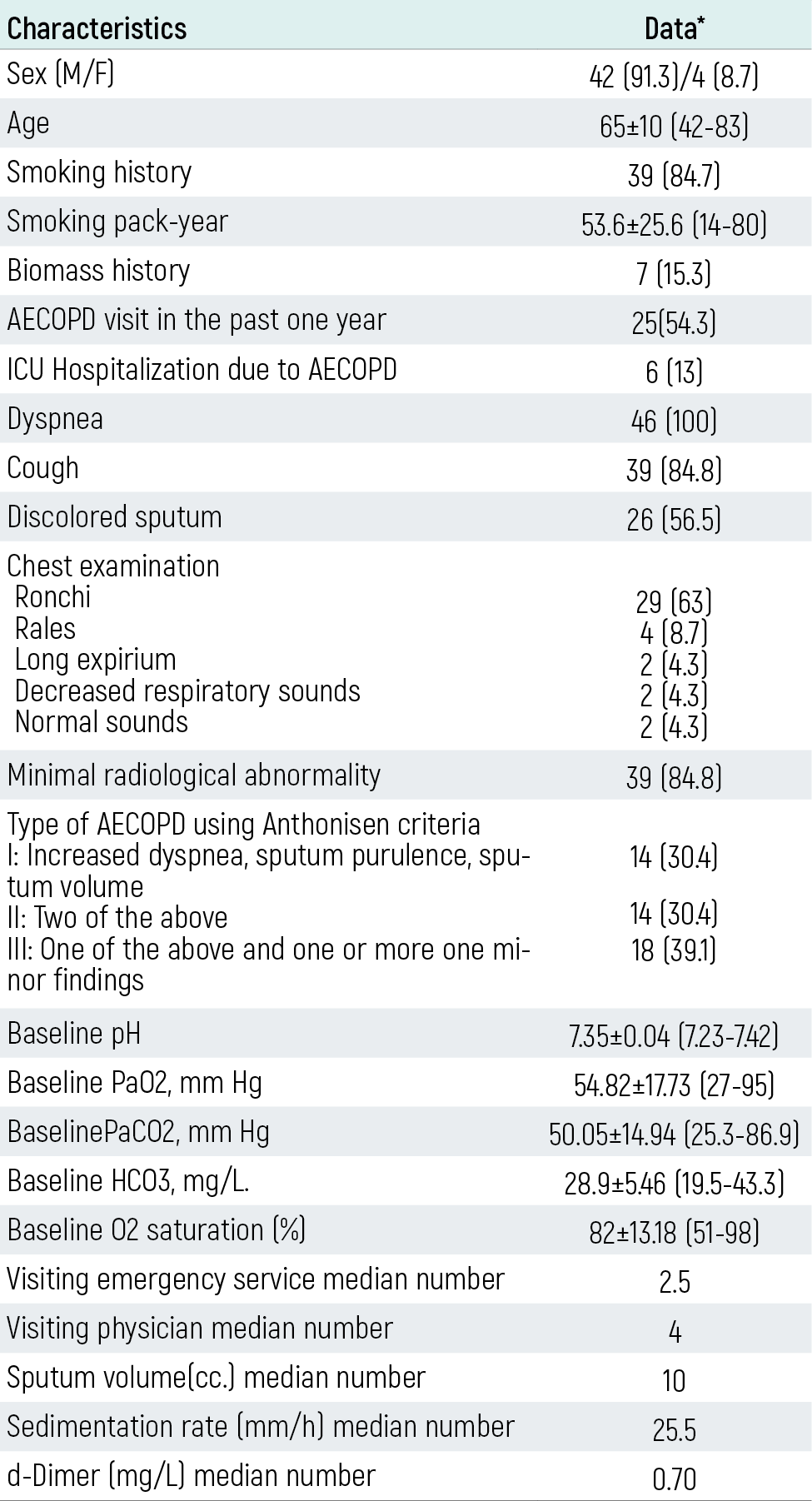
A total of forty-six patients were included in the study: forty-two (91.3%) males and four (8.7%) females. All male patients had a >20 pack-year history of cigarette smoking. Eleven patients (23.9%) had biomass exposure. A total of twenty-five (54.3%) had presented to emergency departments due to acute exacerbation of COPD within the past year, while six (13%) had been admitted to the Intensive Care Unit (ICU) within the past year. Upon admission, all patients (100%) had baseline dyspnea, thirty-nine (84.8%) had a cough, and twenty-six (56.5%) had purulent sputum. In addition, gram staining and nonspecific culture was performed on fourteen patients (30.4%). Physical examination revealed that twenty-nine (63%) had ronchi, five(10.9%) had rales, four (8.7%) had bilateral prolonged expirium, two (4.3%) had decreased respiratory sounds, and two (4.3%) had normal respiratory sounds.
Thirty-nine of the patients (84.8%) had a minimal radiological change during acute exacerbation. Twelve patients (12%) had visited emergency departments service once or twice within the past year, whereas thirty-four (74%) had visited three or more times. Based on Anthonisen criteria, fourteen patients (30.4%) had type I exacerbation, fourteen (30.4%) had type II exacerbation, and eighteen (39.1%) had type III exacerbations. The patients’ characteristics are presented in Table 1.
Median sedimentation rate was 25.5 mm/h, while median d-dimer value was 0.70 mg/L during hospitalization.
Median leukocyte value was 10.500/mm3 on the first day of hospitalization, and 10.250/mm3, 10.050/mm3, 10.300/mm3 on days 3, 7, and 14, respectively.
Mean serum hematocrit value was 43.11%± 5.62% (min:29.1-max:58.8) on the first day of hospitalization, 42.77%± 5.44% (min: %28- max: % 55) on day 3, 42.66%± 5.53% (min:27,4-max: 54.3) on day 7, and 43.12%± 4.36% (min: 35.2- max:51.5) on day 14.
The mean pH of the baseline arterial blood gas was 7.35 ± 0.04 (min:7.23–max: 7.42), mean PaO2 was 54.82 ± 17.73 mmHg (min:27- max:95), mean PaCO2 was 50.05 ± 14.94 mm. Hg (min:25.3- max: 86.9), mean HCO3 was 28.9± 5.46 mmol/L (min:19.5- max:43.3), mean O2 saturation was 82.20%±13.18% (min: 51 - max: 98).
The median CRP level was 3.42 mg/L on the first day of hospitalization, 1.65 mg/L on day 3, 0.73 mg/L on day 7, and 1.15 mg/L on day 14 (Figure 1).
Median BNP values were 97 pg/ml on day 0, 88 pg/ml on day 3, 3.43 pg/ml on day 7, and 2.5 pg/mL on day 14 (Figure 2).
The median copeptin levels was 129.80 pmol/L on the first day, 170.83 pmol/L on day 3, 235.61 pmol/L on day 7, and 338.43 pmol/L on day 14 (Figure 3). A significant difference was found between copeptin levels on day 0, day 3, day 7, and day 14 (p<0.01).
While there was no statistically significant difference between median copeptin levels on day 0 and day 3 (p>0.05), a statistically significant difference was found between the levels on day 0, 7 and 14 (respectively, p<0.05 and p<0.05).
A significant difference was obtained between CRP levels on days 0, 3, 7, and 14 (p<0.001).
A statistically significant difference was found between median CRP values on days 0, 3, 7, and 14 (respectively, p<0.001, p<0.001 and p<0.01).
A significant difference was found between BNP levels on days 0, 3, 7, and 14 (p<0,001).
There was no statistically significant difference between median BNP values on day 0 and day 3 (p>0.05), whereas a statistically significant difference was found between day 0 and day 14 (respectively, p<0.001 and p<0.01).
No statistically significant correlation was found between CRP and copeptin levels (days 0, 3, 7, and 14) (p>0.05). No statistically significant relationship was found between ABG PaO2 and copeptin levels on the first day of hospitalization (p<0.05).
Discussion
Although patients are diagnosed with COPD in clinical practice, no consensus has yet been reached on a gold standard marker for the prognosis and survival of COPD. Leucocytes, CRP and procalcitonin levels associated with the severity of disease are used for diagnosis during acute exacerbation of COPD.3 However, there has been an increase in the previously reported use of Copeptin for prognostic monitoring in clinical settings. This diagnostic use of copeptin, which has been found to be the precursor of AVP, is an important finding particularly in terms of heart failure and septic shock. A recent study has suggested that in comparison to CRP and procalcitonin, copeptin is more effective in determining the survival rate in acute exacerbation of COPD.11
To the best of our knowledge, no other study is currently available determining the course of copeptin measurement in acute exacerbation of COPD. Our aim was to investigate changes in the rate of copeptin at the early stage of exacerbation, investigate a possible relationship between copeptin and CRP in acute exacerbation of COPD, as well as between copeptin and pro-BNP, an indicator of heart failure.
It is known that infections often account for AECOPD. Studies have shown that the Arginine Vasopressin (AVP) level increases in the presence of infections and febrile conditions.13,14 AVP level has also been shown to be associated with hypoxia-related vasoconstriction in severe COPD patients.15,16 AVP is considered to exert adverse inotropic effects on the left ventricle in pulmonary hypertension.17,18,19 AVP has also been shown to increase pulmonary vasoconstrictor response in endotoxemia.15 These findings may be related to increased copeptin levels in poor prognosis of COPD.20
In our study, similar to the study conducted by Stolz et al., no statistically significant relationship was found between copeptin levels with PaO2 levels during hospitalization. On the other hand, copeptin levels were significantly higher in our study, in comparison to other values reported in the literature.
Previous studies have shown increased CRP levels during acute exacerbations of COPD and that these levels were associated with mortality on day 14 and at 6 months.21,22,23
Data has established increased plasma copeptin levels in patients with LRTI and pneumonia.24 Krüger et al reported significantly increased copeptin levels in patients who died on day 28, compared to those of patients who survived pneumonia.25
Studies have also showed that elevated CRP levels provided evidence of side effects in cardiovascular diseases and worsened lung functions in COPD.26,27
CRP may increase rapidly in the presence of infection, and it may decrease quickly once the factor is excluded. However, unlike the study conducted by Stolz et al., we did not find a significant difference between the groups, although CRP levels were higher in Group III compared to Group I based on the Anthonisen criteria (p>0.05).
Copeptin levels were higher in our study in comparison to values reported in the literature. This may be explained by our use of the ELISA technique and its unique scale and cut-off values, in comparison to the approaches applied by others in the literature. In addition, we believe that such a comparison is not appropriate, since the majority of the studies in the literature were conducted by AVP.
To the best of our knowledge, no study is available investigating changes in copeptin levels in the first two weeks of acute exacerbation of COPD.
The results of our study demonstrated that CRP levels increased steadily between day 0 and 14 and the increase was statistically significant (p<0.05). In unison to previous studies, this may be because COPD is a systemic disease, and it affects the whole cardiovascular system.28 An increase in CRP may not be analogous to increased copeptin level in the acute exacerbation of COPD. Furthermore, Stolz et al. also did not find a significant relationship between CRP and copeptin.11
Widespread inflammation occurs in cases of acute exacerbation of COPD. Also, copeptin levels are considered to be increased through stress factors within a short period of time following acute exacerbation of COPD.
Throughout the disease’s progression, complications may be also seen in the clinical presentation of COPD. Chronic airway obstruction leads to hypoxia in the late stages of disease. Increased resistance and, eventually, pulmonary hypertension develop due to pulmonary hypoxic vasoconstriction. The term “cor pulmonale” is used to refer to pulmonary hypertension related to the underlying COPD. Increased pulmonary artery pressure generally results in right ventricular hypertrophy (RVH). In addition, increased jugular venous pressure and hepatomegaly present with cor pulmonale, leading to right heart failure.29
Acute exacerbation of COPD presents with right heart load. As a result, the increase and gradual decrease in BNP levels in our study may be explained by the right heart load in the patients, although heart failure is one of the exclusion criteria at baseline.
In a recent study including 786 patients with chronic heart failure, Neuhold et al. found that in comparison to BNP and NT-proBNP, copeptin was more effective in determining the severity of heart failure in clinical settings. However, these markers were related to each other. On the other hand, no relationship of copeptin to BNP and NT-proBNP was found in the study. The authors found a significant correlation between heart failure and the New York Heart Association (NYHA) Disease Severity Class. In the study, the authors found high copeptin levels to be associated with high mortality risk at 6 months during follow-up.30 In another study, Jochberger et al. demonstrated that serum copeptin and AVP levels returned to normal ranges in patients who underwent cardiac surgery, when cardiovascular functions improved.31 Stoiser and Gegenhuber et al. also reported that increased AVP concentration was associated with poor prognosis and the difference was statistically significant in patients with chronic heart failure. The authors suggested that copeptin was more effective in evaluating prognosis.10,32
Stoiser et al. also suggested that plasma copeptin level measurement and BNP concentration, a standard biomarker of heart failure, were more effective in the evaluation of prognosis.10
A high level of copeptin during baseline may indicate that it functions as an acute phase reactive against hypoxia and microembolism. However, review of the literature revealed no clinical study available to support this finding.
Within the aims of our study, we evaluated copeptin levels for a short period of time at the early stage of the disease in acute exacerbation of COPD. We found that there was a statistically significant difference in copeptin levels between the groups in the short-term. However, the small sample size used in our study should be taken into consideration. The results of our study might demonstrate that COPD is a systemic disease with a variety of phenotypic characteristics and biomarkers used to definitively determine the presence of inflammation, and which has the potential to be influenced by many factors, although acute exacerbation of COPD has similar definitions in the clinical setting.
Conclusion
In conclusion, copeptin, which has similar characteristics to other inflammation markers, is considered to be a novel marker for the prognostic evaluation of acute exacerbation of COPD. We have found no significant correlation between copeptin and WBC, CRP or BNP levels in the short term for AECOPD. However, we did find that copeptin levels did increase gradually from day 1 to day 14. In contrast to previous studies, we conclude that copeptin levels might not be an early inflammation marker in AECOPD. Further large-scale studies with similar phenotypic characteristics are required to define the course and prognostic value of copeptin in AECOPD.
Declarations
Animal and Human Rights Statement
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed Consent
Informed consent was obtained from all participants.
Data Availability
The datasets used and/or analyzed during the current study are not publicly available due to patient privacy reasons but are available from the corresponding author on reasonable request.
Conflict of Interest
The authors declare no conflicts of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the scientific content of the article, including the study design, data collection, analysis and interpretation, manuscript preparation, and approval of the final version of the manuscript.
References
-
Rabe KF, Hurd S, Anzueto A, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2007;176:532-555. doi:10.1164/rccm.200703-456so
-
Evensen AE. Management of COPD exacerbations. Am Fam Physician. 2010;81:607-613.
-
Cazzola M, MacNee W, Martinez FJ, et al. Outcomes for COPD pharmacological trials: from lung function to biomarkers. Eur Respir J. 2008;31:416-469. doi:10.1183/09031936.00099306
-
Le Jemtel TH, Padeletti M, Jelic S. Diagnostic and therapeutic challenges in patients with coexistent chronic obstructive pulmonary disease and chronic heart failure. J Am Coll Cardiol. 2007;49:171-180. doi:10.1016/j.jacc.2006.08.046
-
Burke MA, Cotts WG. Interpretation of B-type natriuretic peptide in cardiac disease and other comorbid conditions. Heart Fail Rev. 2007;12:23-36. doi:10.1007/s10741-007-9002-9
-
Stolz D, Breidthardt T, Christ-Crain M, et al. Use of B-type natriuretic peptide in the risk stratification of acute exacerbations of COPD. Chest. 2008;133:1088-1094. doi:10.1378/chest.07-1959
-
Krismer AC, Dünser MW, Lindner KH, et al. Vasopressin during cardiopulmonary resuscitation and different shock states: a review of the literature. Am J Cardiovasc Drugs. 2006;6:51-68. doi:10.2165/00129784-200606010-00005
-
Mutlu GM, Factor P. Role of vasopressin in the management of septic shock. Intensive Care Med. 2004;30:1276-1291. doi:10.1007/s00134-004-2283-8
-
Landry DW, Oliver JA. The pathogenesis of vasodilatory shock. N Engl J Med. 2001;345:588-595. doi:10.1056/nejmra002709
-
Stoiser B, Mörtl D, Hülsmann M, et al. Copeptin, a fragment of the vasopressin precursor, as a novel predictor of outcome in heart failure. Eur J Clin Invest. 2006;36:771-778. doi:10.1111/j.1365-2362.2006.01724.x
-
Stolz D, Christ-Crain M, Morgenthaler NG, et al. Copeptin, C-reactive protein, and procalcitonin as prognostic biomarkers in acute exacerbation of COPD. Chest. 2007;131:1058-1067. doi:10.1378/chest.06-2336
-
Anthonisen NR, Manfreda J, Warren CP, et al. Antibiotic therapy in exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 1987;106:196-204. doi:10.7326/0003-4819-106-2-196
-
Han DM, Zhang YQ, Bai QX, Chen XQ. Assay of AVP, CRP, and LPS in leukemia. Int J Lab Hematol. 2007;29:185-189. doi:10.1111/j.1751-553x.2006.00839.x
-
Sharples PM, Seckl JR, Human D, et al. Plasma and cerebrospinal fluid arginine vasopressin in patients with and without fever. Arch Dis Child. 1992;67:998-1002. doi:10.1136/adc.67.8.998
-
Westphal M, Stubbe H, Sielenkämper AW, et al. Effects of titrated arginine vasopressin on hemodynamic variables and oxygen transport in healthy and endotoxemic sheep. Crit Care Med. 2003;31:1502-1508. doi:10.1097/01.ccm.0000063042.15272.84
-
Colice GL, Ramirez G. The effect of furosemide during normoxemia and hypoxemia. Am Rev Respir Dis. 1986;133:279-285.
-
Herrera EA, Riquelme RA, Sanhueza EM, et al. Cardiovascular responses to arginine vasopressin blockade during acute hypoxemia in the llama fetus. High Alt Med Biol. 2000;1:175-184. doi:10.1089/15270290050144172
-
Jin HK, Chen YF, Yang RH, et al. Vasopressin lowers pulmonary artery pressure in hypoxic rats by releasing atrial natriuretic peptide. Am J Med Sci. 1989;298:227-236. doi:10.1097/00000441-198910000-00004
-
Jin HK, Yang RH, Chen YF, et al. Hemodynamic effects of arginine vasopressin in rats adapted to chronic hypoxia. J Appl Physiol. 1989;66:151-160. doi:10.1152/jappl.1989.66.1.151
-
Maeder MT, Staub D, Brutsche MH, et al. Copeptin response to clinical maximal exercise tests. Clin Chem. 2010;56:674-686. doi:10.1373/clinchem.2009.136309
-
Dev D, Wallace E, Sankaran R, et al. Value of C-reactive protein measurements in exacerbations of chronic obstructive pulmonary disease. Respir Med. 1998;92:664-667. doi:10.1016/s0954-6111(98)90515-7
-
Hurst JR, Donaldson GC, Perera WR, et al. Use of plasma biomarkers at exacerbation of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2006;174:867-874. doi:10.1164/rccm.200604-506oc
-
Weis N, Almdal T. C-reactive protein—can it be used as a marker of infection in patients with exacerbation of chronic obstructive pulmonary disease? Eur J Intern Med. 2006;17:88-91. doi:10.1016/j.ejim.2005.09.020
-
Müller B, Morgenthaler N, Stolz D, et al. Circulating levels of copeptin, a novel biomarker, in lower respiratory tract infections. Eur J Clin Invest. 2007;37:145-152. doi:10.1111/j.1365-2362.2007.01762.x
-
Krüger S, Ewig S, Kunde J, et al. C-terminal provasopressin (copeptin) in patients with community-acquired pneumonia—Influence of antibiotic pretreatment: results from the German competence network CAPNETZ. J Antimicrob Chemother. 2009;64:159-162.
-
Sin DD, Man SF. Why are patients with chronic obstructive pulmonary disease at increased risk of cardiovascular diseases? The potential role of systemic inflammation in chronic obstructive pulmonary disease. Circulation. 2003;107:1514-1519. doi:10.1161/01.cir.0000056767.69054.b3
-
Gan WQ, Man SF, Sin DD. The interactions between cigarette smoking and reduced lung function on systemic inflammation. Chest. 2005;127:558-564. doi:10.1378/chest.127.2.558
-
Chung KF, Adcock IM. Multifaceted mechanisms in COPD: inflammation, immunity, and tissue repair and destruction. Eur Respir J. 2008;31:1334-1356. doi:10.1183/09031936.00018908
-
Weitzenblum E, Chaouat A. Cor pulmonale. Chron Respir Dis. 2009;6:177-185. doi:10.1177/1479972309104664
-
Neuhold S, Hülsmann M, Strunk G, et al. Comparison of copeptin, B-type natriuretic peptide, and amino-terminal pro–B-type natriuretic peptide in patients with chronic heart failure: prediction of death at different stages of the disease. J Am Coll Cardiol. 2008;52:266-272. doi:10.1016/j.jacc.2008.03.050
-
Jochberger S, Velik-Salchner C, Mayr VD, et al. The vasopressin and copeptin response in patients with vasodilatory shock after cardiac surgery: a prospective, controlled study. Intensive Care Med. 2009;35:489-497. doi:10.1007/s00134-008-1279-1
-
Gegenhuber A, Struck J, Dieplinger B, et al. Comparative evaluation of B-type natriuretic peptide, mid-regional pro–A-type natriuretic peptide, mid-regional pro-adrenomedullin, and copeptin to predict 1-year mortality in patients with acute destabilized heart failure. J Card Fail. 2007;13:42-49. doi:10.1016/j.cardfail.2006.09.004
Figures
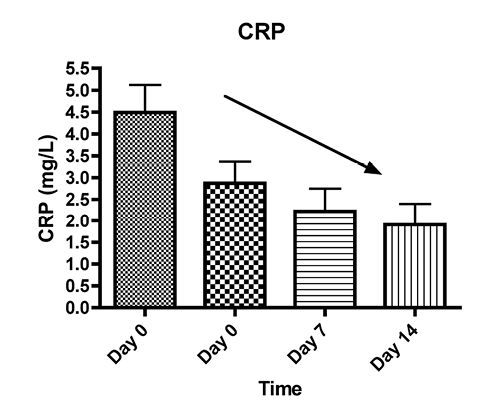
Figure 1. The course of CRP in AECOPD
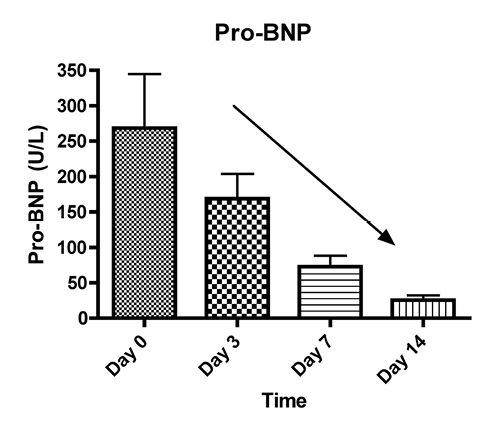
Figure 2. The course of BNP in AECOPD
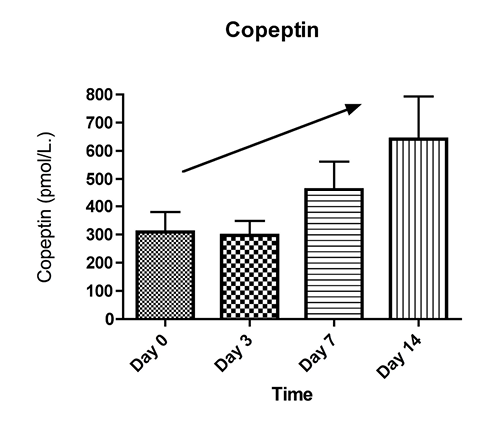
Figure 3. The course of copeptin in AECOPD
Tables
Table 1. Baseline clinical characteristics of 46 patients presenting with AECOPD
* Data are presented an number (%), mean±SD (min-max) or median (IQR). ICU: Intensive Care Unit
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Mehmet Atilla Uysal, Nebibe Bekar, Ersin Demirer, Mehmet Sancak, Veysel Yılmaz. Serum copeptin levels and its relation with other inflammatory markers in acute exacerbation of COPD. Eu Clin Anal Med 2013;1(3):50-55. doi:10.4328/ECAM.21
- Received:
- August 21, 2013
- Accepted:
- September 4, 2013
- Published Online:
- September 4, 2013
- Printed:
- September 4, 2013
