Intraluminal cervical esophageal perforations in rats (experimental study)
Cervical esophageal perforation
Authors
Abstract
AimEsophageal perforation has high morbidity and mortality rates. Morbidity and mortality rates have been decreased slightly by progress in surgical techniques and intensive care conditions. We here aimed to search for the effect of both (a) cervical esophageal perforation from anterior and lateral locations and (b) the safe time interval for repair on mortality and morbidity.
Methods
In our study, we have used 40 rats. The rats were first grouped into anterior and lateral perforation groups; afterwards, these two groups were divided into two subgroups as early (12 h) and late (24 h) repair groups. Perforation was made with a 22-gauge angiocath and repaired in early or late time periods. Before the perforation and treatment, blood samples were collected from the tail vein in order to measure leukocyte levels. Blood sampled before the perforation and while sacrificing the rats—by cardiac puncture—were used to measure IL-10 levels. Contamination status was analyzed and radiological studies were made. Histopathologic examination of the esophagus was made after sacrifice. In order to evaluate the rupture status in treated rats, contrast esophagograms were studied before sacrifice.
ResultsThe groups were evaluated according to the perforation localization, time interval for treatment, white blood cell values revealing the infection, contamination status, IL-10, and fibrosis. We have found a significant difference in white blood cell count and contamination between the rats that were perforated anteriorly and repaired early, and those perforated laterally and repaired in the late time period. Also, we have found a significant difference in contamination between the anterior perforation early repair group and the lateral perforation early and late repair groups. No statistical differences were found within the groups for IL-10, fibrosis, location of the perforation, and the time of the treatment.
ConclusionAs a result, we think that our findings show us that anterior perforation of the cervical esophagus is better tolerated and has a wider safe time interval for treatment.
Keywords
Introduction
Perforation of the esophagus is a surgical emergency because of having high mortality rates.1,2 As the esophagus has no serosa, infections can spread easily and sepsis can establish in a short period of time.3,4,5 Interventional techniques such as tracheostomy, bronchoscopy, nasogastric tube application, videolaryngoscopy, endoscopic esophageal instrumentations, and manipulations in the esophagus for neighboring organs (e.g., Transesophageal echocardiography and ultrasound) are performed much more than before as a result of enhancements in technology and improvements in patient management.
Iatrogenic injuries constitute most of the injuries of the esophagus.2,6,7,8 Most iatrogenic injuries seen in the cervical esophagus are caused by instrumentation.7 In addition, penetrating esophageal injuries3,9 and perforations caused by foreign bodies are commonly seen in the cervical esophagus.10 If injuries of the esophagus are underestimated or overlooked, mediastinitis develops and afterwards the patient dies because of sepsis.4 There are clinical studies evaluating esophageal perforations, but none of them has a consensus on what time it is safe to perform surgery after the injury. We have planned to investigate the safe time interval for surgery.
The cervical esophagus has a special place in esophageal injuries. Because of being the narrowest and the least protected part of the esophagus, the cervical esophagus is more prone to injuries when compared to other segments.
Surgery on time is life-saving in esophageal perforations. The time interval for surgical intervention from the time of injury to the time of treatment is not well established. Knowledge about the time interval has been derived entirely from clinical studies.
This experimental study was established to investigate the direct effect of surgery performed at different time intervals after the perforation, the safe time interval for surgical treatment, and the effect of the perforation site, which has not been studied earlier.
Materials and Methods
GATA ethic committee approval was taken for our study. 40 rats were used (Rattus norvegicus) in our experimental study. The rats were approximately 200-300 g in weight and 4-6 months in age. They were first divided into two categories consisting of 20 rats according to their perforation localization, whether anterior or lateral. These two groups were then divided into 2 subgroups consisting of 10 rats according to their time of treatment. As a result, we have constituted 4 groups each containing 10 rats. While naming these groups, we used the letter “A” for Anterior, “L” for lateral perforation, “E” for treatment after 12 hours (early), and “G” for treatment after 24 hours (late) (Figure 1).
Group “AE” refers to the anterior perforation and early treatment group. Group “AG” refers to the anterior perforation and late treatment group. Group “LE” refers to lateral perforation and early treatment. Group “LG” refers to the lateral perforation and late treatment group. Anterior groups were perforated from the anterior portion, and lateral groups were perforated from the lateral portion of the esophagus. Early groups were treated after 12 hours of injury with primary suturation using 7/0 absorbable material, and late groups were treated after 24 hours with the same material and method.
All the rats were given nothing on the day of operation. Ketamine hydrochloride (90 mg/kg) and xylazine (10 mg/kg) were used as anesthetics, administered parenterally (intramuscular). Before the perforation, we collected blood samples for detecting leukocyte count and IL-10 levels. After collecting the blood samples, we injected the same amount of saline solution intraperitoneally to avoid hemodynamic instability. The rats were stabilized from 4 extremities and the collar region was shaved to avoid contamination. After applying an antiseptic solution and aerosol designed for animals, a vertical 1.5 cm incision was made on the neck.
Blunt dissection was performed to lateralize the collar muscles. Once the trachea was exposed, the carotid sheath was dissected and moved apart from the region to save these important anatomic structures while performing the perforation. Perforation was performed by 22-gauge angiocaths. Two angiocaths were used while performing the perforation. The first one was used as a guide. In order not to make an unwanted perforation to the esophagus, we used this guide. The needle part of the guide angiocath was cut at a distance so that the metal part of it remained within the plastic part (Figure 1). We introduced this guide orally.
When we saw our guide under the microscope in a proper position for perforation, we changed the needle (taken from the second angiocath) to perform the perforation. The perforation was performed anteriorly in the anterior groups and laterally in the lateral groups under the microscope (Figure 2). After the perforation process, the incision was sutured without suturing the esophagus. Depending on the groups, whether early or late, they were reopened for repair of the perforated area. Before the reopening, blood from the tail was sampled to find the leukocyte count. After collecting the blood samples, we injected the same amount of saline solution in order not to create hemodynamic instability.
After opening the skin incision, the perforated area was washed with saline. The wash water was sent for microbiologic analysis to see if there had been any contamination. After washing out, the perforation was sutured with 7/0 absorbable material under a light microscope (Figure 3). The rats were fed orally 24 hours after the operation. Parenteral analgesic and antibiotic treatment were administered for 3 days (amoxicillin 2x0.01, pyralgin 0.05 cc). Before the sacrifice, which was performed after a week, esophagograms were studied to investigate leakage (Figure 4). The rats were sacrificed by cardiac puncture. The collected blood was used to measure IL-10 levels.
After sacrifice, the esophagus was excised for histopathological examination. Fibrosis, granulation tissue formation, and inflammation were examined, alongside investigating the continuity of the lumen in histopathological examination (Figure 5). The collected blood samples were stored in pediatric CBC tubes for leukocyte count. For IL-10 levels, the blood samples were centrifuged and the plasma was used. A specific kit for rat IL-10 was utilized. The first blood samples collected were used as the control group blood. All data were gathered and SPSS 15.0 was used for statistical analysis. Histopathologic, microbiologic, and biochemical data were analyzed with the chi-square test as they are discrete data. Continuous biochemical data were analyzed with the Kruskal-Wallis test. A result of p < 0.05 was accepted as significant.
Results
3 rats were used for the study group in order to establish the perforation model and treatment under a light microscope. 40 rats were used for the experiment groups, which were divided into 4 groups containing 10 rats each. 8 rats were excluded from the study due to various reasons, including severe tissue necrosis (n=3), bronchoconstriction due to tracheal strain (n=2), sudden cardiac arrest (n=1), and major vascular injury (n=2). As a result, the number of rats in each group is shown in the first column of Table 1. The average operation time was 25 \pm 5 minutes.
The histopathologic examination was made by a pathologist under a light microscope blindly. The summary of the histopathologic findings of the experiment can be traced in Table 1.
In order to show the infection in the perforation area, we measured leukocyte values. The rats have a broad range of leukocyte levels and do not have a threshold value for IL-10. Taking into account this knowledge, we planned to measure these values two times and compare them. Control group for leukocytes was formed by the blood samples collected before the perforation. The average value for the control group (8.1 \times 10^3/\text{mm}^3) was compared with the second blood sample values. Second blood sample values are classified as higher or lower than the control group. Table 2 shows the number of rats that have higher or lower values than the control group.
IL-10 levels were measured in the groups from the blood samples collected before the perforation and at the time of sacrifice. The control group was formed by the blood samples collected before the perforation. The mean value for the control group was 78 pg/ml. The values that are above this value are classified as high, and those below this value are low (Table 3).
The microbiological results of the groups are shown in Table 4. All the rats in the lateral perforation groups (LE-LG) had a positive result in their cultures.
Before sacrifice, the esophagograms of the rats were performed to investigate the rupture status. Rupture was shown in 9 rats (81.8%) within 11 histopathologically confirmed ruptured rats. Histopathologically, the existence of rupture is considered a failure in the treatment, and the absence of rupture is considered a success. When we analyzed the groups by rupture, we found no significant result within the groups (p=0.38) (Table 5). However, the absence of rupture was found more frequently in the anterior groups. Rats without rupture were analyzed according to fibrosis; the results were not statistically significant (p=0.77) (Table 6).
When we evaluated the leukocyte count statistically, we found significance between the groups (p=0.03) (Table 7). We found the difference specifically between groups AE and LG. The IL-10 levels were evaluated, and a significant result was not found (p=0.06) (Table 8).
Microbiological analysis that showed contamination had a significant result in statistical analysis (P=0.006) (Table 9). When we examined the groups in pairs, we found that the differences between groups AE-LG and AE-LE were significant.
Discussion
Esophageal perforation has a high rate of mortality and morbidity. Parallel to the advances in the surgical interventions and intensive care units, mortality and morbidity rates have decreased mildly. Beside this decrease, it has a rate of 65% in most of the centers.1,11,12,13,14 The prognosis of the patient is strictly related to patients' general status, type and the location of the injury and time interval between the injury and the time of treatment.8,15,16,17,18,19,20 There are lots of clinical studies showing the importance of early diagnosis and treatment in esophageal perforation. Especially treatment that begins before 24 hours is told to be life saving.8,17,21,22,23,24,25,26 Glatterer and assoc. emphasize the 16 hour for treatment in penetrating injuries of the esophagus.27 A multicenter retrospective study from Asensio and assoc. evaluated the effect of time loss on mortality and morbidity of the patients caused by preoperative evaluation.28 They did not find significant difference on mortality except the complications related to esophagus. Attar and associates found a survival rate of 87% treated within 24 hours whereas 55% treated after 24 hours.21 Pankaj Bhatia and assoc. in their 119 patient retrospective study found the mortality was not associated with the time gap.17 They had found the general status of the patient on admission is much more important for the complications.The reason for different results in different papers for time interval of treatment in esophageal perforation is thought to be related with the patient selection. There can be patients with small perforations admitted after a long time and also large perforations admitted in a short time period.29 Also we think that there can be statistical problems in equal categorization of the patients. In our study, we consider the existence of rupture in histopathologic examination as failure in treatment. When we evaluate the groups by rupture, we have found no statistical difference. The reason for not finding a difference between the groups is thought to be the low number of rats we have. While not finding a significant difference between the groups, when we evaluate Table 1, we can see that the lateral groups (early, late) have a higher number of rupture than the anterior groups.The main reason for having a low number of rupture in anterior groups is thought to be the relationship of esophagus and trachea. This relationship prevents contamination and limits the local inflammation. As a result, we think that localization of the perforation affects the result of the treatment. We have not found a significant difference in time interval by comparing the rupture. We think that if a long time passes for treatment, contamination will disseminate and afterwards inflammation in the tissue will improve leading to sepsis that will affect the treatment.We have found a statistical difference between groups in contamination (p=0.006). The difference was found between the anterior early group and lateral early and late group. This shows us that the perforations from the anterior aspect are less contaminated. Less contamination brings fewer complications, good surgical outcomes and good results in late treatments. Parallel to the contamination, leukocyte values in lateral groups are significantly higher than the anterior groups (p=0.03). This shows us the anterior perforations have a low level of contamination thus leading to less inflammation. These results reveal that perforation site is important in esophageal perforation.There is correlation in the papers on the studies performed for the diagnosis of esophageal perforations. Direct graphies are used first following a contrast esophagograms afterwards. Esophagograms can be repeated or computed tomograms can be used if there is a suspect in diagnosis. Onat and assoc. mentioned that the best way to evaluate the perforation is the contrast esophagograms by computed tomography.3 We have seen rupture in most of the rats (81.8%) by esophagograms which is confirmed histopathologically. Contrast esophagograms can be used in the diagnosis of esophageal perforations safely but negative results should not be thought as intact lumen.The last period in tissue remodeling is fibrosis. Low fibrosis formation is associated with good tissue elasticity and function. Thus, we evaluated the fibrosis formation in unruptured rats. We have not found a statistically difference between the groups. All the groups had a mild fibrosis but more in anterior groups. This shows us primary suturing is healed with mild fibrosis that leads to minimal loss of functionality of the esophagus. IL-10 is an anti-inflammatory cytokine that is primarily produced from monocytes and less produced by leukocytes. Researches revealed that IL-10 is the main immune modulator cytokine in gastrointestinal system and recombinant IL-10 suppresses inflammation in bowels in Crohn's disease.30 We have not found significance between groups. But when we consider Table 3 and Table 8, lateral late group has more high levels and mean value. We think that the higher IL-10 in lateral late group reveals the more severe inflammation in the lateral late group.There are lots of papers related with esophageal perforations, however there is no consensus on the treatment of it. Non operative treatment is commonly used in iatrogenic injuries and cervical region perforations.13 When the conservative treatment fails, surgery is then performed. This time delay prepares complications. For this reason, non operative treatment should be chosen properly to avoid such complications.31,32 The mainstay of the treatment is to avoid contamination and infection, rebuilding of feeding, maintenance of gastrointestinal continuity.33 Cervical esophageal perforations and thoracal perforations that are confined by the pleural folds can be treated conservatively.34 Gupta and Asensio had declared the main treatment in esophageal perforations is early performed surgery in their papers.28,35 Tsalis and co-workers informed in their case report that the management protocol plays an important role in the mortality rate.36 We used the primary suturing protocol in our study. Primary suturing is commonly used in the treatment of esophageal perforations. In lateral perforations of the esophagus, we have found that lack of supportive tissue results in contamination of the mediastinum. Before performing the lateral perforation in our study, carotid sheath dissection is carried out to avoid the injury to the vessels. This dissection can be a factor for spreading the infection. Also, a laceration to the vessels can lead to a hematoma in the region that compresses the vascular structures or esophagus. And also esophagovascular fistulization may occur. These complications should be kept in mind also.Before performing the anterior perforation, we dissect esophagus from trachea in order not to injury trachea. In the perforations of the anterior esophagus, tracheal involvement must be strictly evaluated. If tracheal injury is excluded, then conservative treatment should be promptly used. Esophageal perforation is a highly mortal and morbid injury. There is no standard treatment algorithm in perforations. Patients should be evaluated individually. In our study, we have found that anterior perforations of the esophagus is better tolerated when treated with primary suturation. Also, we have shown in our study that late recognized anterior perforations of the esophagus has a better result than early recognized lateral perforations. For this reason, we think that anterior perforations of the esophagus is better tolerated than the lateral perforations.
Conclusion
In our study, we have found that anterior perforations of the esophagus is better tolerated and has a wider range of time interval. We have not found a difference in time interval but we think that increased time is associated with high rates of fail in treatment that will lead high mortality rates and complications. Our study will be supported by the experimental or clinical studies in the future.
Declarations
Animal and Human Rights Statement
All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Informed Consent
Not applicable.
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding
None.
Scientific Responsibility Statement
The authors declare that they are responsible for the scientific content of the article, including the study design, data collection, analysis and interpretation, manuscript preparation, and approval of the final version of the manuscript.
References
-
Kuppusamy MK, Felisky C, Kozarek RA, et al. Impact of endoscopic assessment and treatment on operative and nonoperative management of acute oesophageal perforation. Br J Surg. 2011;98(6):818-824. doi:10.1002/bjs.7437
-
Eroğlu A, Aydın Y, Aksoy M. Özofagus yaralanmaları. Toraks. 2011.
-
Onat S, Ulku R, Cigdem KM, et al. Factors affecting the outcome of surgically treated noniatrogenic traumatic cervical esophageal perforation: 28 years’ experience at a single center. J Cardiothorac Surg. 2010;5:46. doi:10.1186/1749-8090-5-46
-
Özçelik C, İnci İ. Özofagus yaralanmaları. In: Özofagus hastalıklarının tıbbi ve cerrahi tedavisi. Özlem Grafik Matbaacılık; 2002:77-108.
-
Panieri E, Millar AJ, Rode H, et al. Iatrogenic esophageal perforation in children: patterns of injury, presentation, management, and outcome. J Pediatr Surg. 1996;31(7):890-895. doi:10.1016/s0022-3468(96)90404-2
-
Wright CD. Özofagus perforasyonunun tedavisi. In: Çağırıcı U, Turhan K, eds. Erişkin göğüs cerrahisi. Nobel Matbaacılık; 2011:353-360.
-
Paul S, Chang MY. Künt ve penetran özofagus yaralanmaları. In: Çağırıcı U, Turhan K, eds. Erişkin göğüs cerrahisi. Nobel Matbaacılık; 2011:361-372.
-
Brinster CJ, Singhal S, Lee L, et al. Evolving options in the management of esophageal perforation. Ann Thorac Surg. 2004;77:1475-1483. doi:10.1016/j.athoracsur.2003.08.037
-
Wu JT, Mattox KL, Wall MJ Jr. Esophageal perforations: new perspectives and treatment paradigms. J Trauma. 2007;63(5):1173-1184. doi:10.1097/ta.0b013e31805c0dd4
-
Nandi P, Ong GB. Foreign body in the oesophagus: review of 2394 cases. Br J Surg. 1978;65:5-9. doi:10.1002/bjs.1800650103
-
Huber-Lang M, Henne-Bruns D, Schmitz B, Wuerl P. Esophageal perforation: principles of diagnosis and surgical management. Surg Today. 2006;36:332-340. doi:10.1007/s00595-005-3158-5
-
Port JL, Kent MS, Korst RJ, et al. Thoracic esophageal perforations: a decade of experience. Ann Thorac Surg. 2003;75:1071-1074. doi:10.1016/s0003-4975(02)04650-7
-
Vogel SB, Rout WR, Martin TD, Abbitt PL. Esophageal perforation in adults: aggressive conservative treatment lowers morbidity and mortality. Ann Surg. 2005;241:1016-1021. doi:10.1097/01.sla.0000164183.91898.74
-
Kim-Deobald J, Kozarek RA. Esophageal perforation: an 8-year review of a multispecialty clinic’s experience. Am J Gastroenterol. 1992;87:1112-1119.
-
Okten I, Cangir AK, Ozdemir N, et al. Management of esophageal perforation. Surg Today. 2001;21:36-39.
-
Kiev J, Amendola M, Bouhaidar D, et al. A management algorithm for esophageal perforation. Am J Surg. 2007;194:103-106. doi:10.1016/j.amjsurg.2006.07.024
-
Bhatia P, Fortin D, Inculet RI, Malthaner RA. Current concepts in the management of esophageal perforations: a 27-year Canadian experience. Ann Thorac Surg. 2011;92(1):209-215. doi:10.1016/j.athoracsur.2011.03.131
-
Eroglu A, Can Kürkçüoglu I, Karaoglanoglu N, et al. Esophageal perforation: the importance of early diagnosis and primary repair. Dis Esophagus. 2004;17:91-94.
-
Kotsis L, Kostic S, Zubovitz K. Multimodality treatment of esophageal disruptions. Chest. 1997;112:1304-1309. doi:10.1378/chest.112.5.1304
-
Johnsson E, Lundell L, Leidman B. Sealing of esophageal perforation or ruptures with expandable metallic stents: a prospective controlled study. Dis Esophagus. 2005;18:262-266. doi:10.1111/j.1442-2050.2005.00476.x
-
Attar S, Hankins JR, Suter CM, et al. Esophageal perforation: a therapeutic challenge. Ann Thorac Surg. 1990;50(1):45-49. doi:10.1016/0003-4975(90)90082-h
-
Wright CD, Mathisen DJ, Wain JC, et al. Reinforced primary repair of thoracic esophageal perforation. Ann Thorac Surg. 1995;60:245-249. doi:10.1016/0003-4975(95)00377-w
-
Salo JA, Isolauri JO, Heikkila LJ, et al. Management of delayed esophageal perforation with mediastinal sepsis: esophagectomy or primary repair? J Thorac Cardiovasc Surg. 1993;106:1088-1091. doi:10.1016/s0022-5223(19)33982-0
-
White RK, Morris DM. Diagnosis and management of esophageal perforations. Am Surg. 1992;58:112-119.
-
Reeder LB, DeFilippi VJ, Ferguson MK. Current results of therapy for esophageal perforation. Am J Surg. 1995;169(6):615-617. doi:10.1016/s0002-9610(99)80232-3
-
Muir AD, White J, McGuigan JA, et al. Treatment and outcomes of oesophageal perforation in a tertiary referral centre. Eur J Cardiothorac Surg. 2003;23(5):799-804. doi:10.1016/s1010-7940(03)00050-2
-
Glatterer MS Jr, Toon RS, Ellestad C, et al. Management of blunt and penetrating external esophageal trauma. J Trauma. 1985;25(8):784-792. doi:10.1097/00005373-198508000-00009
-
Asensio JA, Chahwan S, Forno W, et al. Penetrating esophageal injuries: multicenter study of the American Association for the Surgery of Trauma. J Trauma. 2001;50(2):289-296. doi:10.1097/00005373-200102000-00015
-
Shaker H, Elsayed H, Whittle I, et al. The influence of the “golden 24-hour rule” on the prognosis of oesophageal perforation in the modern era. Eur J Cardiothorac Surg. 2010;38(2):216-222. doi:10.1016/j.ejcts.2010.01.030
-
Braat H, Rottiers P, Hommes DW, et al. A phase I trial with transgenic bacteria expressing interleukin-10 in Crohn disease. Clin Gastroenterol Hepatol. 2006;4(6):754-759. doi:10.1016/j.cgh.2006.03.028
-
Griffin SM, Lamb PJ, Shenfine J, et al. Spontaneous rupture of the oesophagus. Br J Surg. 2008;95(9):1115-1120. doi:10.1002/bjs.6294
-
Altorjay A, Kiss J, Vörös A, Bohák A. Nonoperative management of esophageal perforations: is it justified? Ann Surg. 1997;225(4):415-421. doi:10.1097/00000658-199704000-00011
-
Jones WG, Ginsberg RJ. Esophageal perforation: a continuing challenge. Ann Thorac Surg. 1992;53:534-543. doi:10.1016/0003-4975(92)90294-e
-
Kanowitz A, Markovchick V. Oesophageal and diaphragmatic trauma. In: Rosen P, ed. Emergency medicine: concepts and clinical practice. 4th ed. Mosby; 1998:546-548.
-
Gupta NM, Kaman L. Personal management of 57 consecutive patients with esophageal perforation. Am J Surg. 2004;187(1):58-63. doi:10.1016/j.amjsurg.2002.11.004
-
Tsalis K, Blouhos K, Kapetanos D, et al. Conservative management for an esophageal perforation in a patient presented with delayed diagnosis: a case report and review of the literature. Cases J. 2009;2:6784. doi:10.4076/1757-1626-2-6784
Figures

Figure 1. The Distrubition of the groups
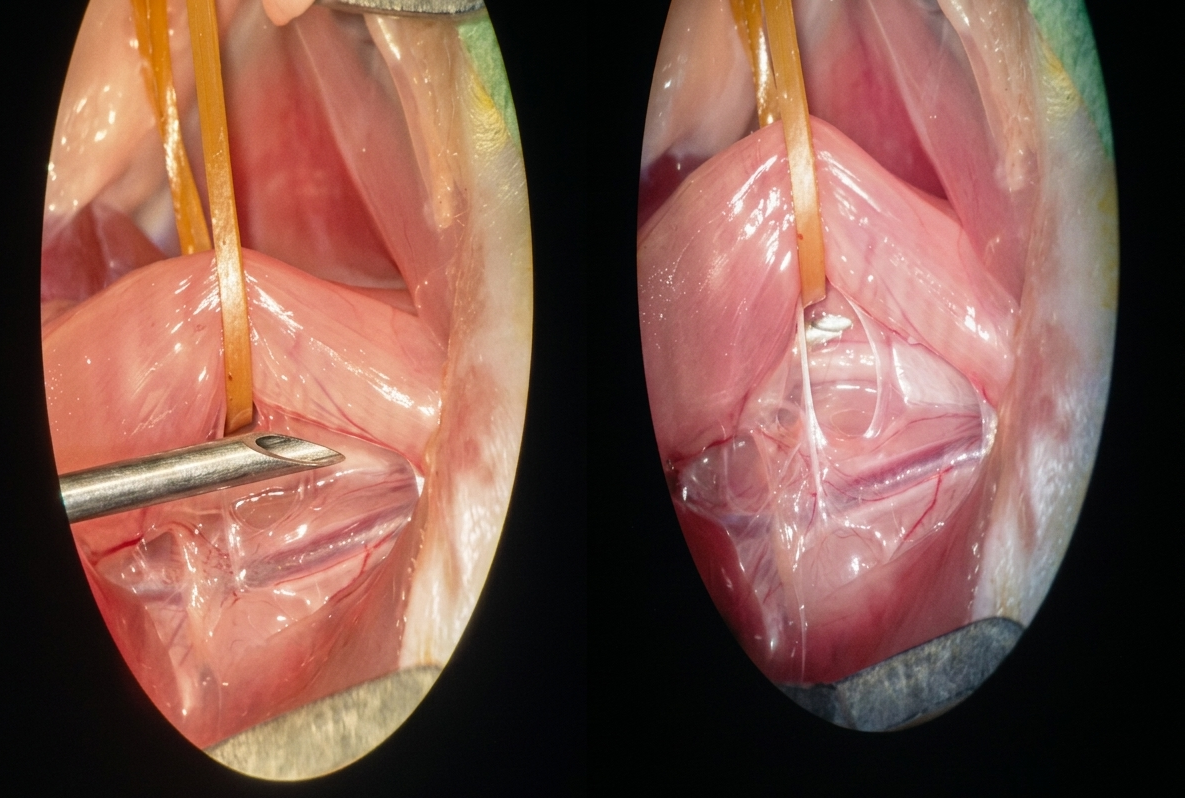
Figure 2. The angiocaths used for the perforation

Figure 3. Perforation of the esophageus from the lateral and anterior wall
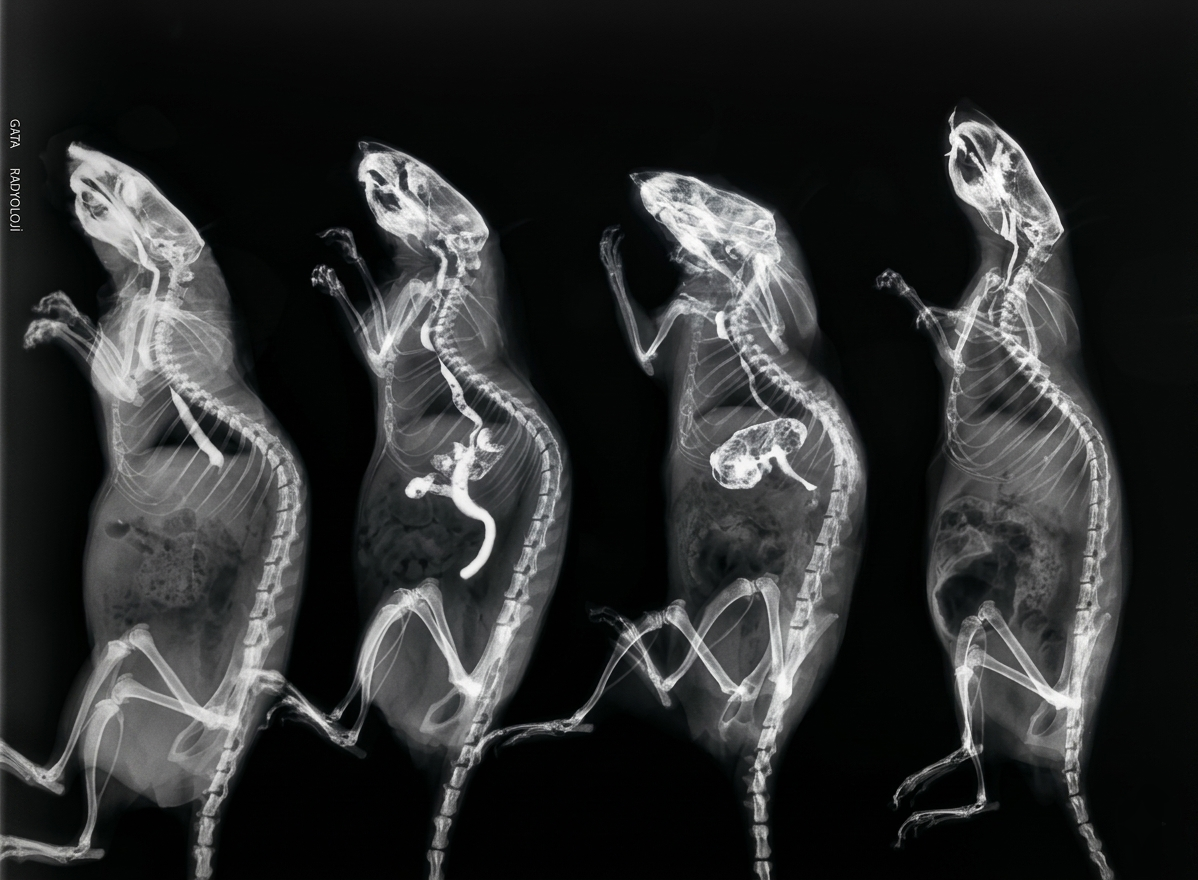
Figure 4. Primary suturation of the perforation site
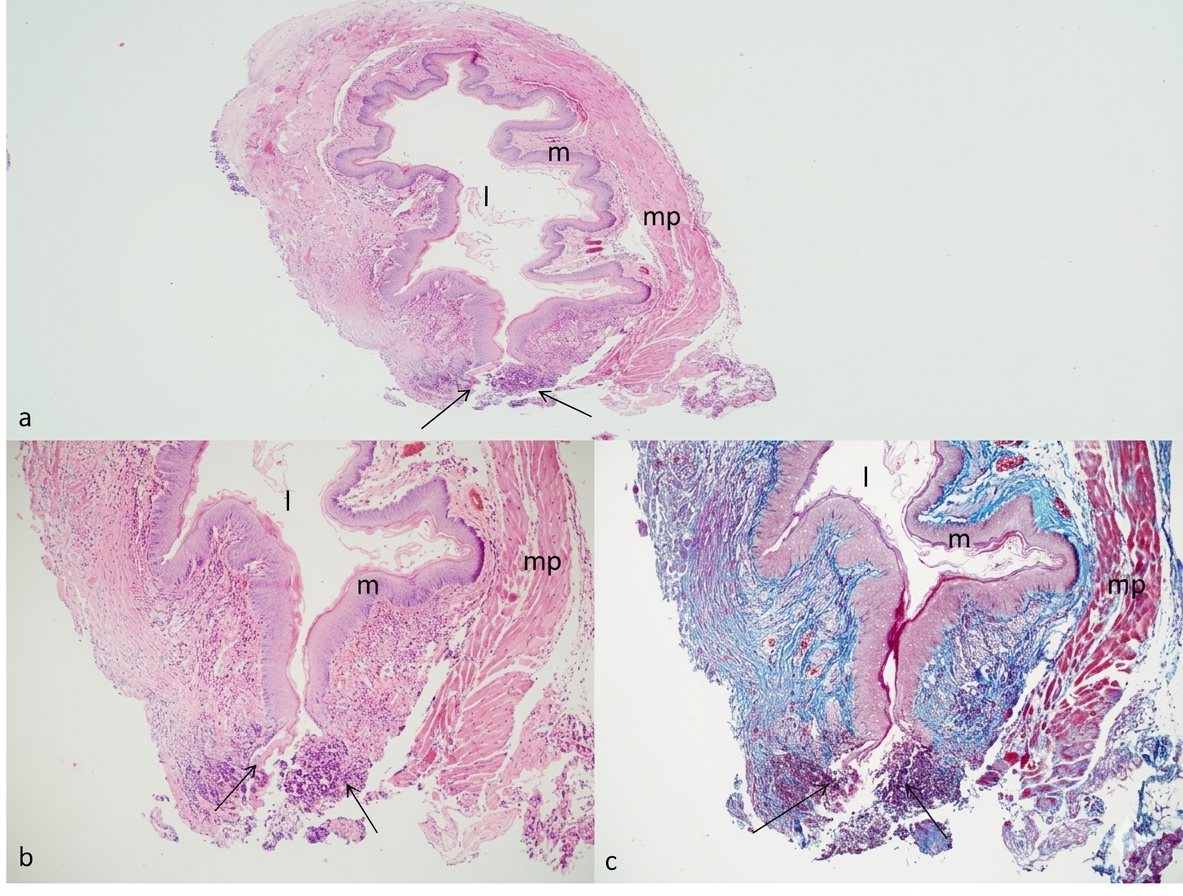
Figure 5. Esophagograms of the rats
Additional Information
Publisher’s Note
Bayrakol MP remains neutral with regard to jurisdictional and institutional claims.
Rights and Permissions
About This Article
How to Cite This Article
Sezai Çubuk, Orhan Yücel, Alper Gözübüyük, Hasan Çaylak, Armağan Günal, Elmir Memmedov, Özlem Öztürk, Sedat Gürkök. Intraluminal cervical esophageal perforations in rats (experimental study). Eu Clin Anal Med 2013;1(1):4-9. doi:10.4328/ECAM.5
- Received:
- March 9, 2013
- Accepted:
- April 1, 2013
- Published Online:
- April 4, 2013
- Printed:
- April 4, 2013
